
Iron transition metal Chemistry iron(II) Fe2+ iron(III) Fe3+ complexes ions ligand substitution redox chemical reactions principal oxidation states +2 +3 extraction GCE AS A2 IB A level inorganic chemistry revision notes
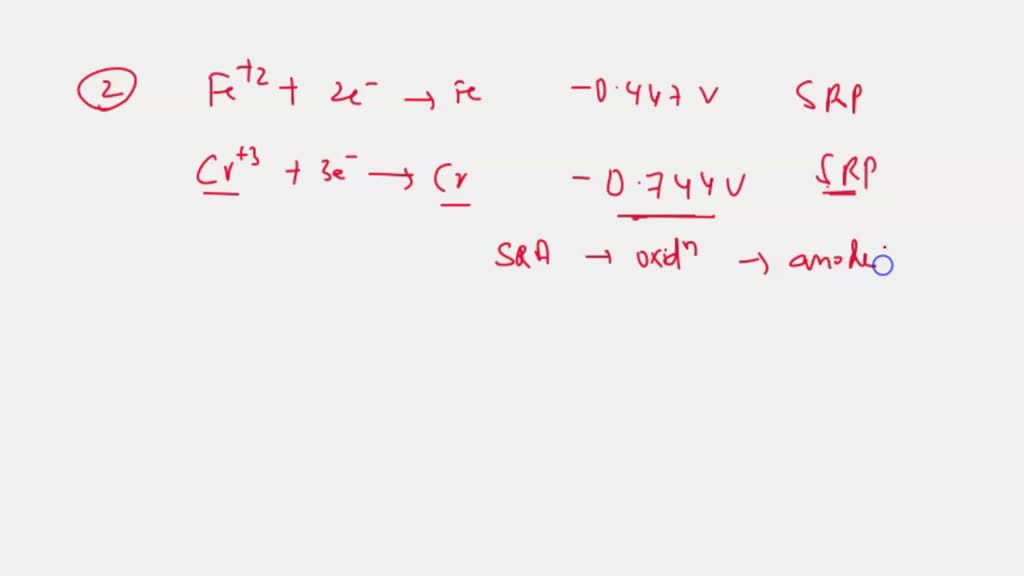
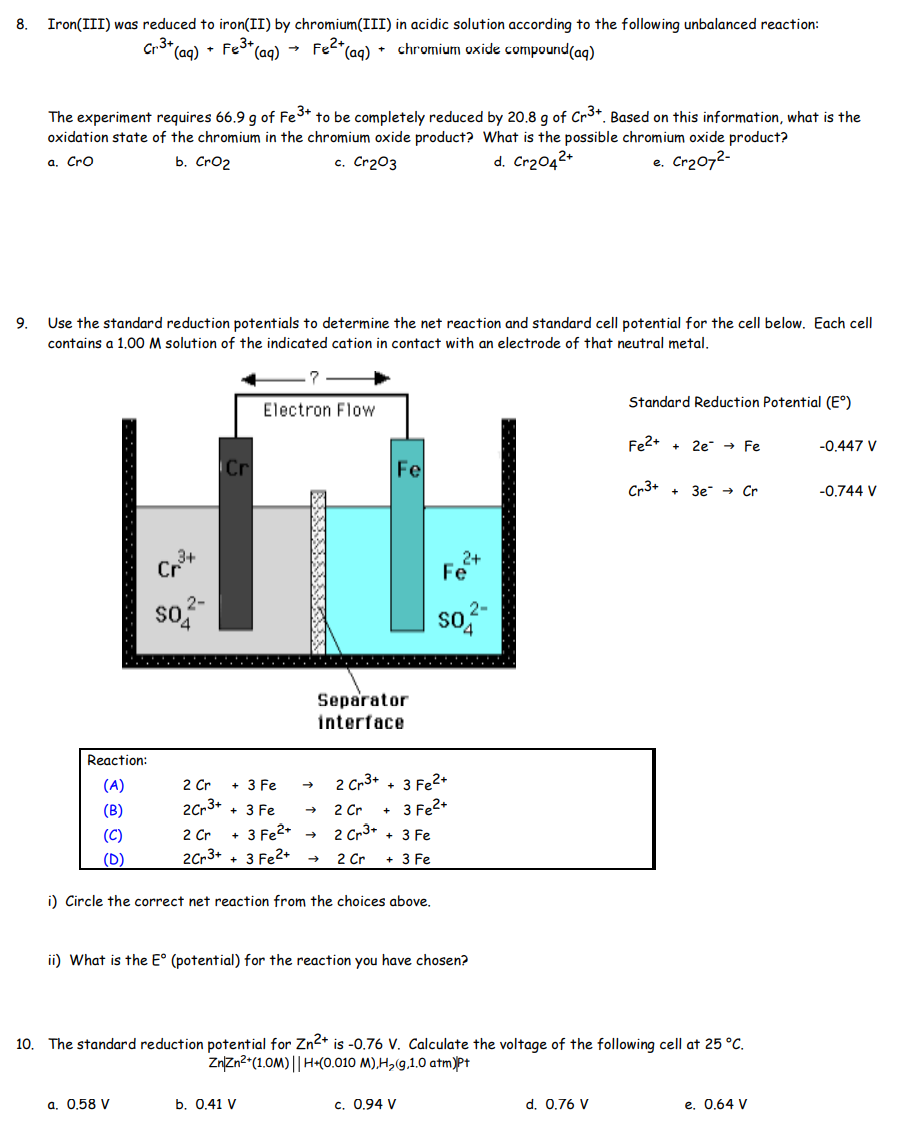
SOLVED: Iron(III) was reduced to iron(II) by chromium(III) in acidic solution according to the following unbalanced reaction: Cr3+(aq) + Fe3+(aq) ⟷ Fe2+(aq) + chromium oxide compound(aq) The experiment requires 66.9 g of

Photo-catalytic selectivity of anthranilic acid over iron oxide incorporated titania nanoparticles: Influence of the Fe2+/Fe3+ ratio of iron oxide - ScienceDirect

Electrochemical Analysis of Changes in Iron Oxide Reducibility during Abiotic Ferrihydrite Transformation into Goethite and Magnetite | Environmental Science & Technology

PDF) Phase Transformation of Iron Oxide Nanoparticles by Varying the Molar Ratio of Fe2+: Fe3+ | Samaneh Alibeigi - Academia.edu
If Fe2+ and Fe3+ are present in Oxide of Iron Fe0.78O0.96 .Then Calculate Number of These Isotopes in Iron Oxide and Their

Determining Magnetite/Maghemite Composition and Core–Shell Nanostructure from Magnetization Curve for Iron Oxide Nanoparticles | The Journal of Physical Chemistry C
![The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11051-011-0495-8/MediaObjects/11051_2011_495_Fig6_HTML.gif)
The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink

Chemical Origin of Sodium Phosphate Interactions on Iron and Iron Oxide Surfaces by First Principle Calculations | The Journal of Physical Chemistry C

Manetite, which has the formula Fe(2)O(3) is comprised or iron(II) oxide and iron (III) oxide. What is the ratio of iron(II) ions to iron(III) ions in magnetiude.

Figure 3 from First-Principles Fe L2,3-Edge and O K-Edge XANES and XMCD Spectra for Iron Oxides. | Semantic Scholar

Droop, G. T. R. (1987) A general equation for estimating Fe3+ concentrations in ferromagnesian silicates and oxides from microprobe analyses, using stoichiometric criteria. Mineralogical Magazine, 51 (361). 431-435 doi:10.1180/minmag.1987.051.361.10
![PDF) The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system PDF) The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system](https://i1.rgstatic.net/publication/225853005_The_effect_of_Fe3Fe2_molar_ratio_and_iron_salts_concentration_on_the_properties_of_superparamagnetic_iron_oxide_nanoparticles_in_the_waterethanoltoluene_system/links/56c1317208ae44da37fc2171/largepreview.png)
PDF) The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system

Figure 1 from Electronic spectra of Fe 3 + oxides and oxide hydroxides in the near IR to near UV | Semantic Scholar






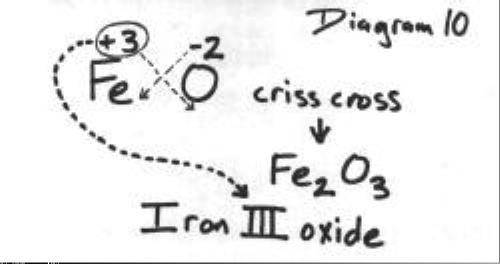

![A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0304885317309411-gr4.jpg)
