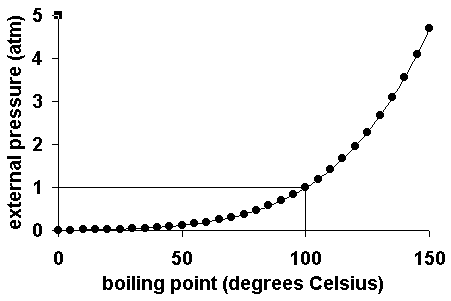
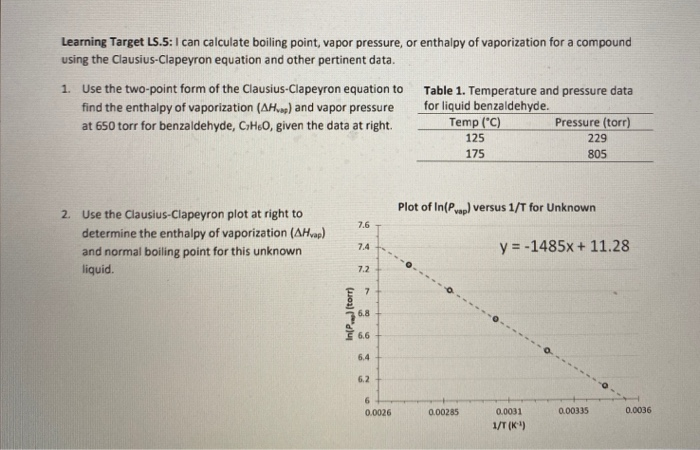
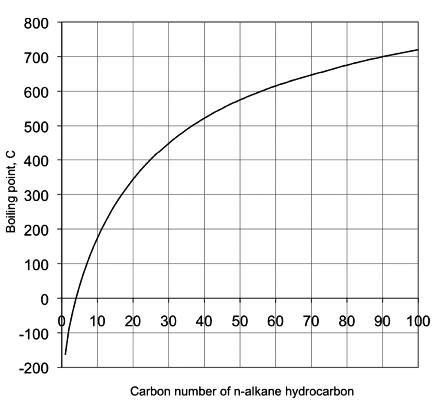
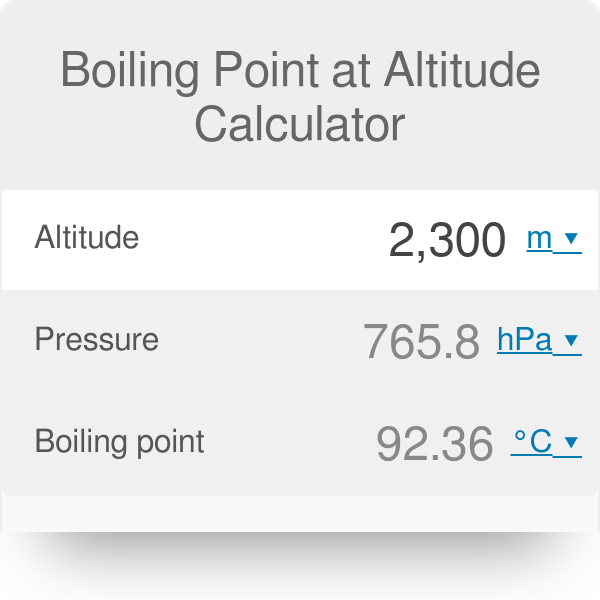
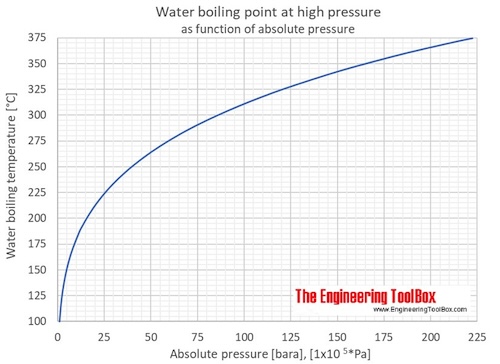
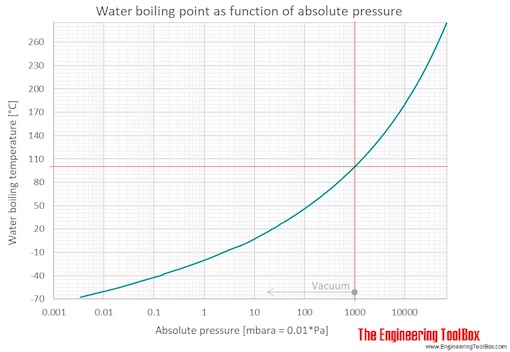
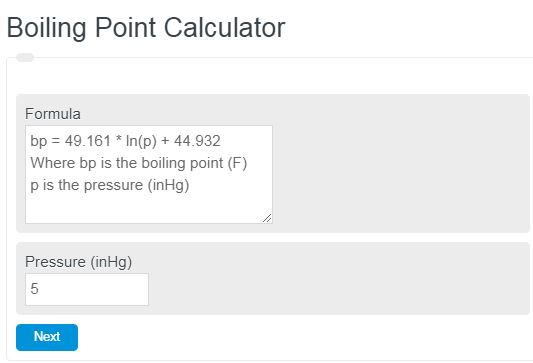
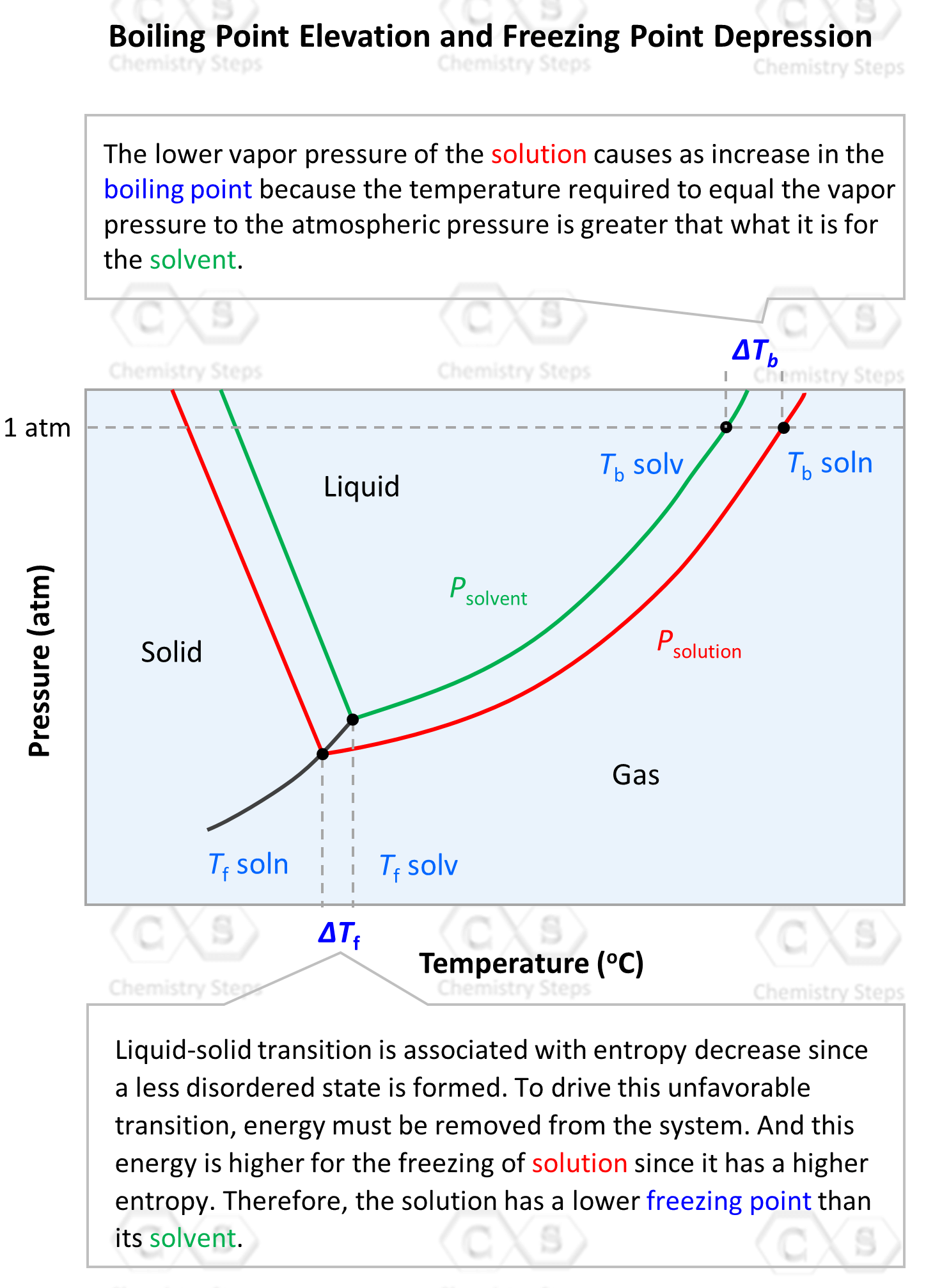
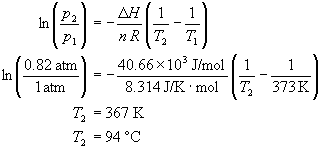How does one calculate the boiling point of water at different pressures without a steam table? - Quora

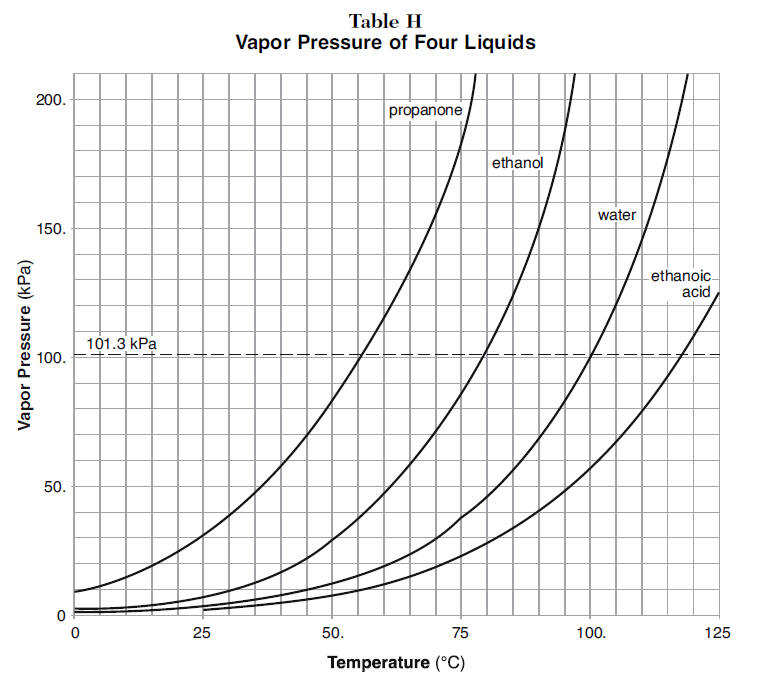
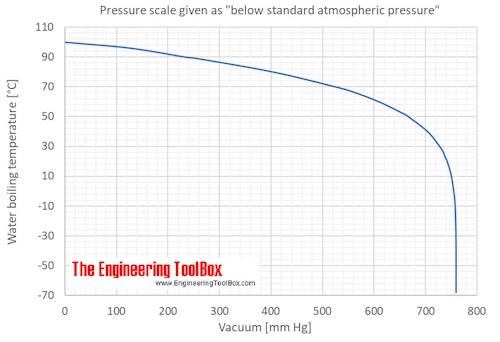
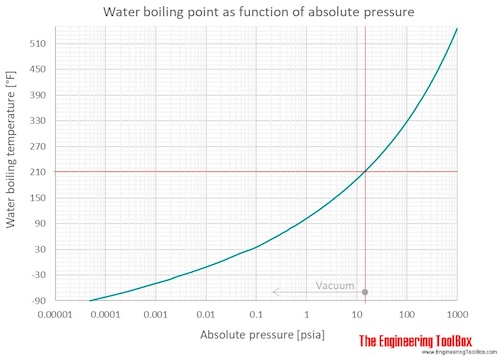
Calculate the boiling point of water at 24 torr pressure. The average Hvap. over the temperature range is 10.12 kcal mol^-1 . Will all the water form gaseous state if placed in